San Diego Regenerative Medicine Institute
Human Embryonic Stem Cell Therapy --- Unleashing the Power of Life Sciences


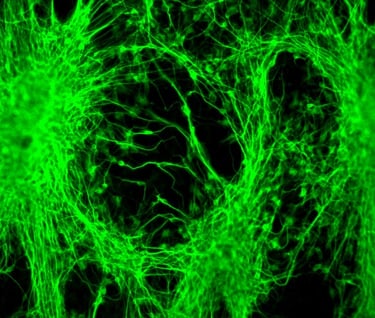
Transforming the Future of Medicine
San Diego Regenerative Medicine Institute (SDRMI), a nonprofit independent biomedical research institute, was founded to facilitate the transition of human stem cell research towards stem cell therapy to provide the next generation of cell-based therapeutic solutions for unmet medical challenges in world-wide major health problems. SDRMI is committed to the leading position in pluripotent human embryonic stem cell (hESC) research and cell-based regenerative medicine by developing groundbreaking hESC technology platforms and innovative hESC-based regenerative medicine approaches.


Revolutionizing Patient Care with Human Embryonic Stem Cells
The successful derivation of human embryonic stem cell (hESC) lines from the in vitro fertilization (IVF) leftover embryos is considered as one of the major breakthroughs of the 20th century life sciences. In regenerative medicine, hESC research holds huge promise for treating major human diseases that have been challenging for traditional medicine, such as a wide range of incurable or hitherto untreatable neurological and heart diseases. Millions of people are pinning their hopes on hESC research. Despite these devastating and life-threatening diseases are leading causes of death or permanent disability, yet, there is no effective treatment or drug available. The limit capacity of cardiomyocytes (the mature contracting heart muscle cells) of the heart as well as neuron circuitries of the brain/spinal-cord for self-repair constitutes a significant challenge to traditional medicine for tissue and function restoration in seeking cures for those serious diseases and conditions. To date, the need to restore vital tissue and function for a wide range of incurable or hitherto untreatable neurological and heart diseases remains a daunting challenge to the conventional mode of drug development.
Although stem cell therapy represents a promising regenerative medicine approach closest to provide a cure for those diseases, demonstrating stem cell production at the scale and product purity adequate to heal the damaged or lost tissues that have naturally limited capacity for repair, such as the human heart and brain, has been a big challenge for traditional adult stem cell sources or products, including so-called induced pluripotent adult/stem cells (iPSC) that are in fact adult cells reprogrammed with oncogenes or cancer cells, and another adult stem cell Ponzi scheme or scam. As neurological and cardiovascular diseases incur exorbitant costs on the healthcare system worldwide, there is a strong focus on translating hESC research innovations to provide potentially life-saving treatments or cures for these major health problems.


Breakthrough Human Embryonic Stem Cell Technology Platforms and Regenerative Medicine Innovations
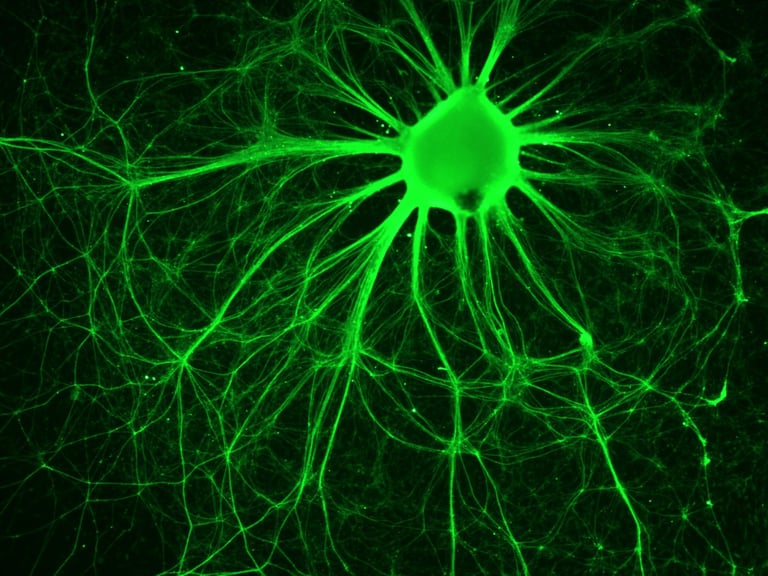
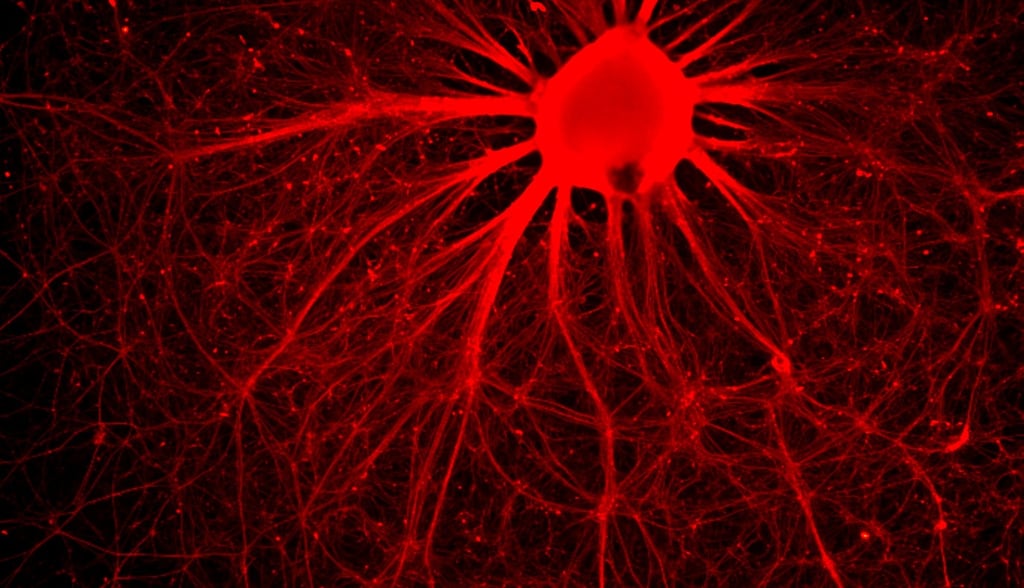
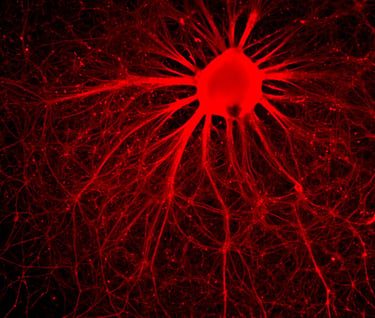
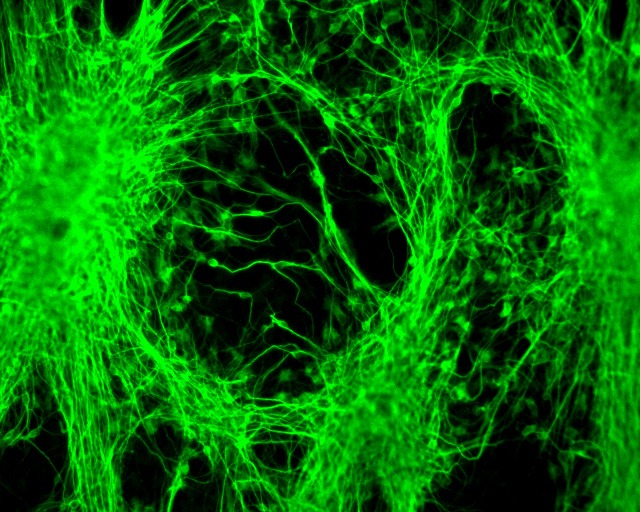
SDRMI’s breakthrough hESC technology platform – PluriXcel -- allows well-controlled, highly efficient, direct conversion of non-functional clinical-grade hESC at the pluripotent stage by small molecule induction into a large supply of functional human neuronal progenitor cells (Xcel-hNuP) or human cardiac precursor cells (Xcel-CardP) as novel, safe, and effective regenerative medicine advanced therapy (RMAT) products for neuron-circuitry or heart-muscle regeneration, overcoming the major bottlenecks in the regenerative medicine market. Our PluriXcel Platform is a game-changing enabling technology to provide RMAT products in large quantity and high quality with adequate cellular capacity to regenerate the neuron circuitry and the contractile heart muscle, ensuring high degrees of efficacy and safety of the hESC-derived therapeutic products, thus robust clinical benefit leading to therapies. It not only constitutes clinically representative progresses in both human neuronal and cardiac therapeutic products for treating a wide range of incurable or hitherto untreatable neurological and cardiovascular diseases, but also offers manufacturing innovation for production scale-up and creation of replacement tissue or organ products. Our breakthrough innovations present hESC as a novel, advanced therapeutic strategy for a wide range of incurable or hitherto untreatable neurological and heart diseases, having tremendous impact on economy, health, future medicine, and patient care.
Human Embryonic Stem Cell Patents
USPTO# 8,716,017, “Technologies, Methods, & Products of Small Molecule-Directed Tissue & Organ Regeneration from Human Pluripotent Stem Cells”. First U.S. patent for large-scale production of clinical-grade high quality human embryonic stem cell lines and their neuronal cell therapy products.
USPTO# 9,428,731, “Technologies, Methods, & Products of Small Molecule-Directed Tissue & Organ Regeneration from Human Pluripotent Stem Cells”. First U.S. patent for large-scale production of clinical-grade high quality human embryonic stem cell lines and their heart muscle cell therapy products.